FST JOURNAL
Critical Minerals
DOI: https://www.doi.org/10.53289/BJDE2755
Critical Minerals and battery technology
Professor Emma Kendrick

Professor of Energy Materials, School of Metallurgy and Materials, University of Birmingham and co-lead of the Energy Materials Group (EMG). Her research focuses on the design and development of sustainable battery technologies and chemistries. Materials lifecycle, from raw material to component, manufacturing, lifetime, and recycling. Prior to academia, she led innovations in the battery industry, as Chief Technologist in Energy Storage at SHARP Laboratories of Europe Ltd (SLE) and as Lead Scientist of two lithium-ion battery SMEs, Fife Batteries Ltd and Surion Energy Ltd.
Summary:
- Classification of materials as "critical" varies by region
- In a battery, mixed lithium, manganese, cobalt and nickel oxides are carefully engineered and then formulated into an electrode coating onto metal foils.
- Most lithium mining occurs in Australia, and nearly all of it is shipped to China for processing. Once processed, the lithium is then distributed worldwide
- Cobalt is typically a secondary element. Around 70% of the world's cobalt is sourced from the Democratic Republic of Congo (DRC), and most of it is shipped to China where the refining takes place before worldwide distribution
- To enhance sustainability, we could reduce the reliance on critical materials, explore the reuse of batteries and their components, and focus on recycling. One possibility is to increase the energy density of our devices.
I would like to present a case study focused on technology and the understanding of critical materials within that technology, specifically batteries. As a battery scientist, I aim to illustrate how innovations can impact the use of critical materials and how we might reduce the quantity of these materials in our battery technologies.
Battery science
Let us start by discussing the basic structure of a battery. For those who may not know, a battery consists of two main components: the anode and the cathode. The anode is typically made of graphite, which is classified as a critical material or mineral. The cathode, on the other hand, is commonly composed of a mixed metal oxide that includes lithium, nickel, cobalt, and manganese. It's important to note that the classification of these materials as "critical" varies by region. For example, in Europe, lithium, manganese, and cobalt are considered critical, while nickel is categorised as a strategic material due to its low supply chain risk. In a battery, these materials are carefully engineered and coated onto metals. The anode, made of graphite, is connected to a copper current collector, while the cathode, formed from mixed metal oxide, is attached to an aluminum current collector. These materials are precisely designed to create spherical particles that pack together efficiently, maximising energy storage in a small space while allowing enough space for electrolytes to flow. The electrolyte is responsible for transporting lithium ions between the anode and cathode. During battery operation, lithium ions move from the graphite anode to the cathode, while electrons flow through the external circuit.
Materials
I will now focus on a case study involving specific materials. Firstly, lithium, particularly in the context of the European critical materials list, which has been established for some time. In 2024, we will begin analysing data from the recent UK critical materials and minerals list. Globally, two primary regions produce lithium. Australia accounts for over 50% of the world's lithium supply, while the remainder primarily comes from South America, specifically from salt flats or brines. In these regions, brines are pumped from salt lakes, allowing the water to evaporate, which leaves behind concentrated lithium deposits that are then exported and refined. Australia extracts lithium from a rock called spodumene. Most of the lithium mining occurs in Australia, and nearly all of it is shipped to China for processing. Once processed, the lithium is then distributed worldwide.
The story of cobalt, nickel, and copper is quite different. Cobalt is typically a secondary element obtained from various ores, such as copper or nickel ore. In fact, around 70% of the world's cobalt is sourced from the Democratic Republic of Congo (DRC), and most of it is shipped to China where the refining takes place before it reaches the rest of the world. When we discussed nickel as a strategic material, we noted that its supply chain risk is significantly lower. Europe has a considerable number of nickel refineries and some nickel and copper mines, which helps to mitigate the supply chain risks associated with these materials.
Sustainability
So, what can we do to enhance the sustainability of these technologies? One approach is to reduce the reliance on critical materials, explore the reuse of batteries and their components, and focus on recycling. This aligns with the well-known waste hierarchy for sustainability. To minimise material usage, we can first increase the energy density of our devices. By improving energy density, we require less material overall. Additionally, increasing the lifespan of battery technologies can play a crucial role in sustainability since longer-lasting materials mean a smaller initial value chain. To achieve this, battery manufacturing can incorporate substitutions. For instance, we can consider substituting lithium with sodium or cobalt with iron. Sodium is abundant, including sources like soda ash in the UK and seawater, which is globally available. However, substituting lithium with sodium comes with trade-offs; sodium is heavier and larger, which reduces the overall energy density and average voltage. On the other hand, iron is far more abundant and globally available compared to cobalt, and it is also significantly cheaper. In battery technology, substitutions can be made by replacing lithium with sodium and adjusting the electrolyte, as well as swapping graphite for hard carbon. Additionally, we can substitute copper for aluminum and replace cobalt with iron. Currently, there is a new battery technology being developed in China that focuses on these substitutions, particularly for use in lower-range vehicles.
Recycling in relation to batteries
If you take a Nissan LEAF battery cell and disassemble it to recover its materials, a typical method would involve shredding. This process essentially destroys the intricate engineering that went into creating the battery with its carefully crafted components. After shredding, the challenge is to reclaim and purify the resulting materials. If we could disassemble battery cells instead of shredding them, we could separate the materials with less waste, making the process potentially more economically viable. This raises an important question about innovation: can we design new battery technologies with disassembly in mind? By designing for easier disassembly, we can enhance material recovery while minimizing waste. When discussing substitution in battery technologies, we can compare different types of chemistries.
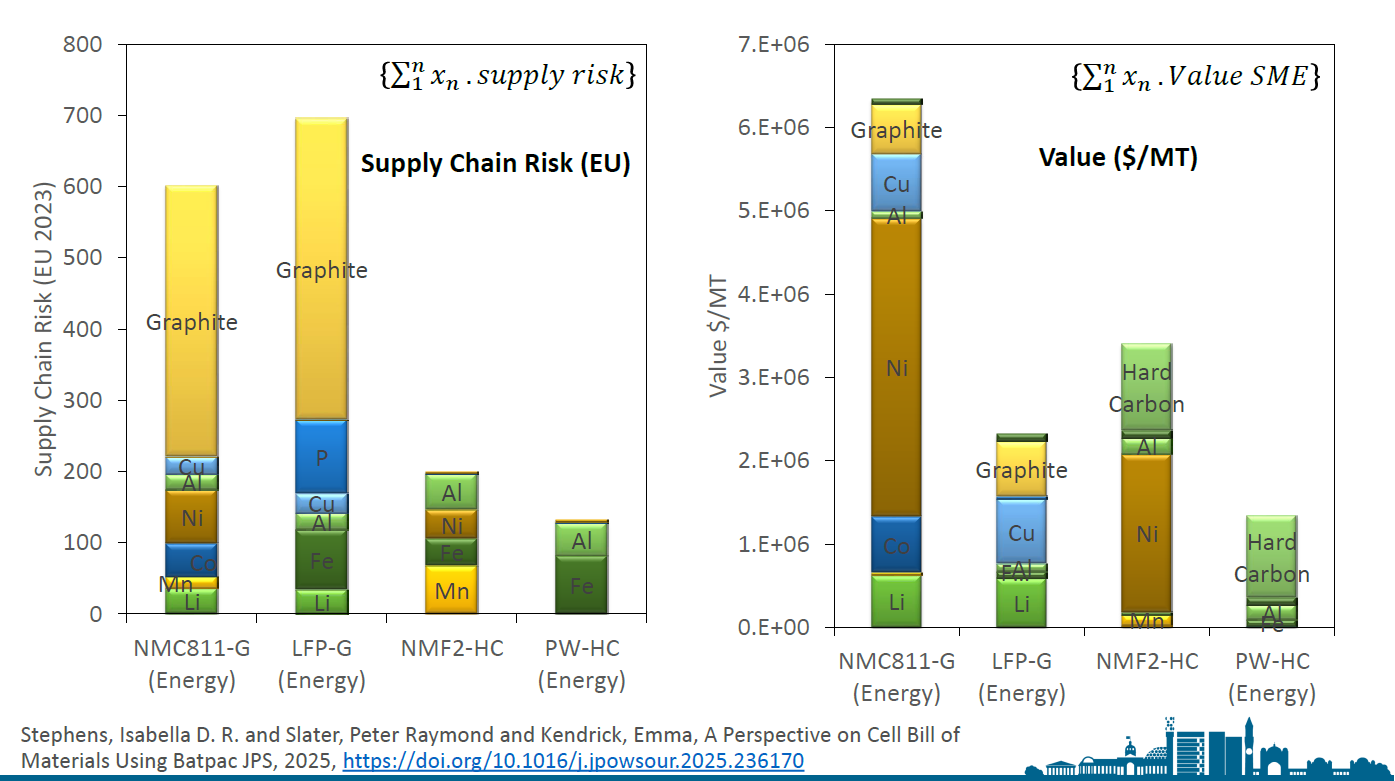
We can begin to compare the quantities of materials used in different technologies and how these impact both the cost and the supply chain risk associated with these minerals. For instance, in lithium-ion technologies, graphite poses a significant supply chain risk, as 99.9% of battery-grade graphite comes from a single country: yes, that's China. Additionally, when we look at the value of materials, nickel, cobalt, and copper have the highest values. Therefore, there is a strong economic incentive to recycle these materials. On the other hand, if we consider lithium iron phosphate and some sodium-ion technologies, which contain lower-value components, we need to find a way to reclaim those materials as well. Ultimately, we must ask ourselves: what happens at the end of a battery's life? If there is no economically viable solution for recovering these materials, what will become of them?
